 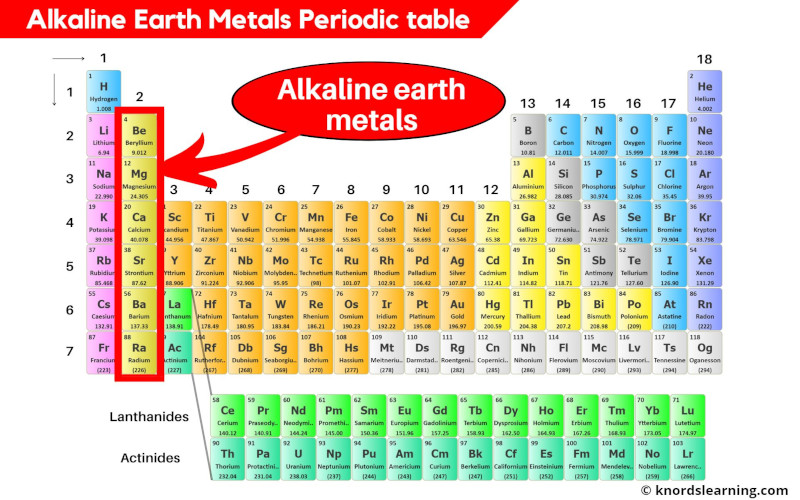
The transition elements are elements where the d orbitals (groups 3–11 on the periodic table) are filling, and the inner transition metals are the elements where the f orbitals are filling. The representative elements are elements where the s and p orbitals are filling. It is possible to divide elements into groups according to their electron configurations. The primary focus of this section will be the application of periodicity to the representative metals. We begin this section by examining the behaviors of representative metals in relation to their positions in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
